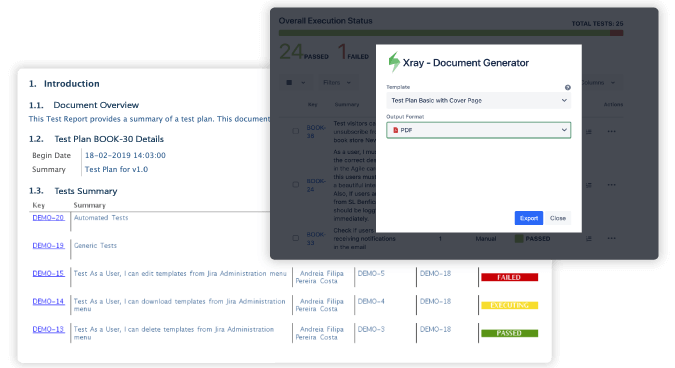
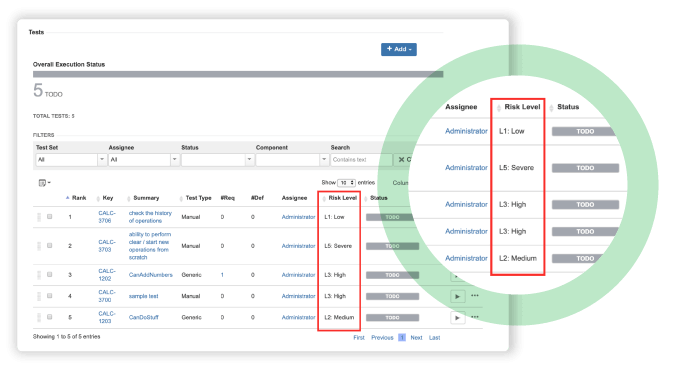
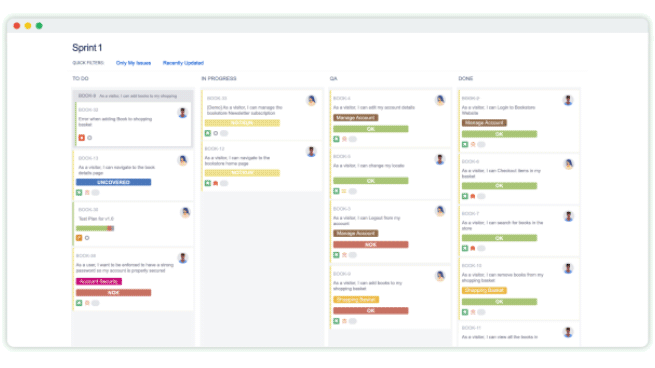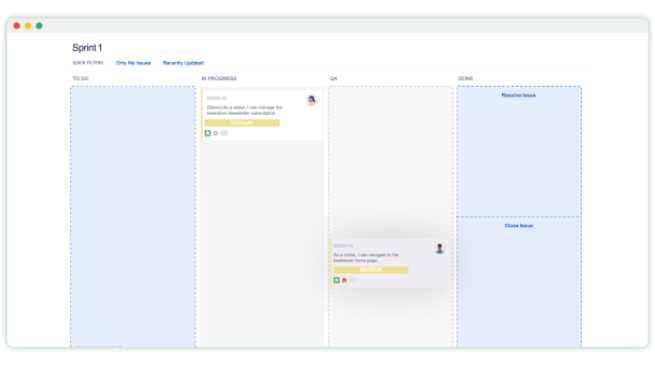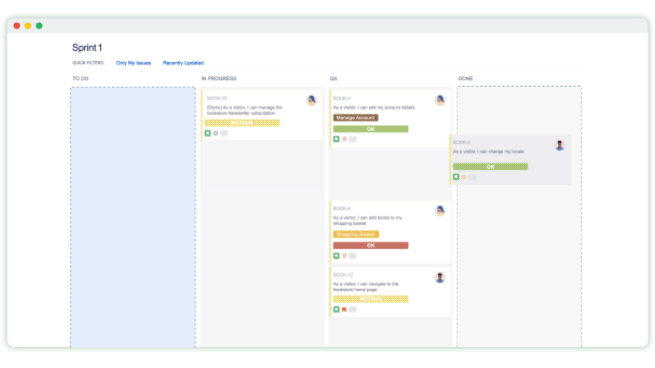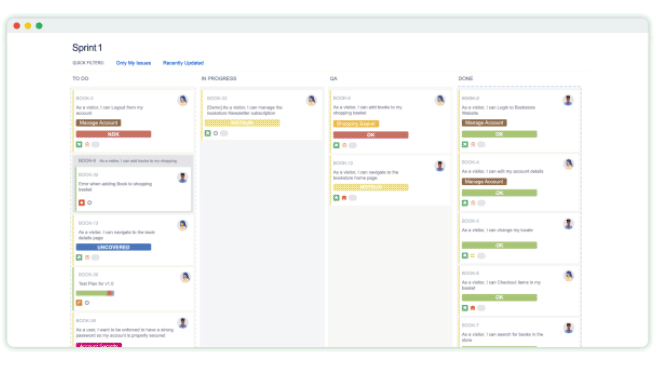
How a medical device manufacturer meets mission-critical regulations with Xray
Learn how a medical device manufacturer uses Xray to ensure full requirements traceability, optimize test reporting time and meet critical FDA and ISO regulations.
READ NOW